There’s been recurring themes in much of the research we’ve undertaken while finding new Twilight Beasts for this blog. Overkill, as discussed by Ross in his harrowing blog on the Steller Sea Cow is one of them; environmental changes caused by agricultural expansion is another. A peculiar theme which I’ve noted is the perpetuation of creatures within folklore, as distorted memories. This was evident in the indigenous legends involving Castoroides ohioensis, and pretty likely with the ghoulish modern urban myth of the Mothman being a continuance of the memory of Ornimegalonyx.
I know a little of what the twilight world of dreams and myths are like, for as a child growing up at the height of the euphemistically named ‘Troubles’ in Belfast, my imagination was populated by entities who inhabited my personal refugia, the Ulster Museum. Egyptian mummies and Megaloceros skeletons were not frightening; instead they were benign beings waiting for re-fleshing, their bony state merely an impermanence which tethered them to the same time and place as me.
Perhaps not surprisingly, I eventually became an archaeologist, with specific interests in environmental issues, and how we work within our landscapes and livestock. I’m most fascinated by humans interaction with animals, how we share our spaces and our lives, how the creatures we domesticate become imbued with status – in fact, this is part of my thesis-to-be, focusing on horses in the Iron Age! As I’ve researched various Pleistocene creatures here I’ve realised the creatures that manage to avoid domestication also become smattered with humanities dream-dust, becoming either folkloric angel or demon; big bad wolf or Arthurian white hart.
The question of course has been how direct that folk memory is. It’s usually speculated that early peoples found bones, and hunters recognised them as being similar to creatures they were familiar with, only bigger. The stories kick in afterwards, with no tribal ancestor ever having actually witnessed the creatures themselves.
I was, therefore, pretty interested by the headlines circulating social media regarding new dates for the last Siberian Megaloceros: the research cautiously suggest Mesolithic/early Neolithic humans would have overlapped with the Giant Deer.

A wonderful illustration of a proud male Megaloceros giganteus standing protectively over his mate. Those enormous antlers could grow longer than 12 foot! (Art by Tabitha Paterson)
Some websites reporting this new research indicated Bronze Age dates for the last of the Giant Deer – that is, they were around until only 5000 years before present (BP). This would have been a game-changer in every aspect for both palaeontologist and archaeologist. Within minutes I admit I had accessed my university library website, had Quaternary Science Reviews open and was reading that paper! Sadly, the Bronze Age dates aren’t there, but it is a high quality, well researched paper with excellent chronological results and perhaps an indication of some interesting archaeological possibilities.
This experienced research team developed substantially on the chronology of Giant Deer (Megaloceros giganteus) extinction created during the 1990s. It’s likely that the Giant Deer ceased to exist in Europe around 12,600 BP (calibrated), right on the Younger Dryas, although there’s also evidence suggesting some persisted in the British Isles until the warmer Littletonian phase around 11,500 BP (calibrated). It’s been accepted that there were small, isolated pockets of these majestic creatures surviving in Western Siberia until around 7700 BP, well within the Siberian Mesolithic/Neolithic transitional phase of human settlement.
One of the authors, Yaroslav Kuzmin who works at the Sobolev Institute of Geology and Mineralogy in Russia, has worked on numerous archaeological sites in Siberia, and has contributed greatly to a better understanding of Siberian prehistory. As regions go, Siberia is an incredibly important place, with its Ural mountain boundaries being the home grounds of the Yamna and Sintashta cultures of the Bronze Age. To understand the deep prehistory of the area is to get the prequel of the makings of those societies – and our own. The samples of Megaloceros bone, therefore, were picked from sites which have provided evidence of long-term human use. A sample of antler was found at a deliberate deposition in a grave of the 3rd millennium BC, in the south-western Siberian region of Sopka. Likewise, a maxilla of Megaloceros, with only one molar missing, was found in a burial context at the persistent burial ground of Preobrazhenka, also south-western Siberia. Other samples were taken from the Ust-Tushama and Sosnovy Tushamsk sites in the Angara river basin, in eastern Siberia.
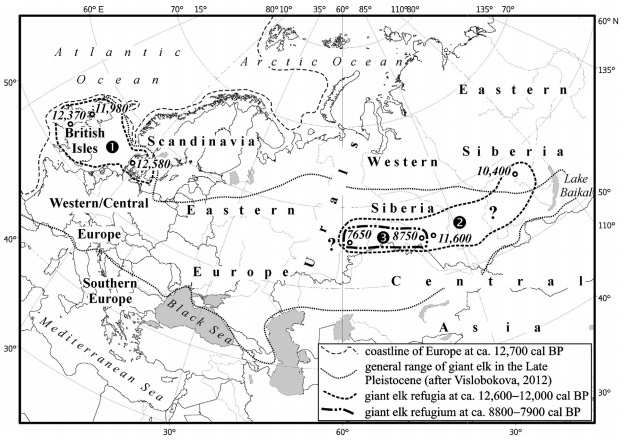
The long expanse of the Siberian landscape. Illustrating the range of the Giant Deer localities (image from van der Plicht et al. 2015)
The radiocarbon dates are robust – from the reading of the article and the footnotes I have little doubt that the quality of the Megaloceros dental collagen used to produce the dates was enviably good. When modelled beside previous work by Stuart et al (2004), the picture shows an ever shrinking territory of this Pleistocene giant. By the end of the Younger Dryas, around 11,600 BP, the Trans-Ural populations still extended into eastern Siberia, but the next 2000 years resulted in their final decline in a restricted refugial zone of western Siberia, with radiocarbon dates suggesting that extinction occurred sometime after 7600 BP, on the boundary of steppe and mountain. It’s hard to say, however, if this is terminus post or ante quem. It’s clear there’s a lot of new research waiting in the wings, reinforcing what exciting times we live in for all things Pleistocene!
Now, this is a very strong hypothesis and it certainly means that western Siberian hunters of the Mesolithic period would most certainly have caught glimpses of the Giant Deer. It’s likely they hunted them too – food in these harsh environments would have been less abundant than warmer, luscious plains. Imagine the stories they would tell of these rare creatures, and how they passed into myth. When later societies found the huge antlers, and remembered the old, old stories of those eyewitnesses, it is no wonder that those tines were considered to be gifts worth placing within the graves of the palaeometal cultures of the 3rd millennium BC, linking them with the otherworldly land of the ancestors. After all, the dearly departed were making the journey to the same place as those giant Pleistocene monarchs of the steppes.
The steppes nestle at the foot of each side of the Ural mountain range. Presumably humans had long watched herds of wild horse, and various deer ( Megaloceros perhaps?) and ached to gallop across that landscape. Botai, in Kazakhstan, is just south of the final Megaloceros refugial zone; it is the site which offers an early date for the domestication of the horse, around 6000yrs BP. That domestication opened fresh vistas for the nomadic people of the region, new places to winter with their herds of livestock. It also opened trade routes and networks, which in turn created more sophisticated societies which ran in tandem with the development of metalwork.
Around 5000 years ago, the Bronze Age saw changes in burials – it was not sufficient to place a treasured heirloom of a prehistoric deer antler within a grave any more. Chieftains were interred with often exquisitely worked goods to see them into the afterlife. The Sintashta burials are elaborate, with horses and deer placed in graves with chariots and gold objects. The more mobile steppe tribes of early kurgan cultures again linked deer and horses in their burials, as noted by the ubiquitous J. P Mallory, academic par excellence. This link seems to persist from those early Bronze Age times right through to the Eurasian Iron Age. C 500 BC.
Many have heard of the Pazyryk ‘Ice Princess‘, in Siberia. Her exceptionally well preserved burial chamber was found in an area of the Altai called Ukok, known evocatively as the ‘pastures of heaven’. She was a special person to her tribe of Iron Age pastoralists, and archaeology has developed a pretty strong understanding of her early death, likely due to breast cancer. Her semi-mummified body is tattooed with fabulous creatures, which may hint at her holding a shamanic status rather than a royal one. The image of the great spreading antlers of bellowing deer is a recurring image on both her body art and her grave goods.
She is not alone in her embellishments. The great deer with huge antlers is a theme occurring in many Pazyryk graves. Rudenko and Gryaznov worked on many of these kurgan graves through the 1950s and 1960s, and it was not just the humans who were identified with the fantastical deer. Horses which had been sacrificed and placed within the graves were often masked with headgear which made them look like deer. Fabulous creatures now in death, hybrids of the flying deer depicted on Altai standing stones, and the familiar horse, ready to gallop through heaven where the ancestors of man, horse and deer would know never-ending freedom.

A rather wonderful, if slightly bizarre, horse decorated as a deer. A Giant Deer perhaps? (Image from Rudenko, 1970)
Dr Ruth Carden, doyenne of all things Megaloceros, pondered if the deer depicted in these images and masks could be in fact the Siberian Red Deer. There’s a possibility of this, but the horns of the depicted creatures are so ornate, it’s hard to say. They are ornate and stylised , resembling the way a dreamer- or a child- would depict the Giant Irish Deer. Memory and myths are persistent things as we have seen from other Pleistocene creatures which were still half remembered from even earlier phases of human history. So, just for fun, let’s propose a scenario – and it’s only one of many which we can speculate on.
The research by van der Plicht’s team found samples of Megaloceros bones, which were from around 8900 to 8600 BP cal, as deliberate deposits in 5000 year old graves of the Siberian Neolithic. The memory of giant, lordly deer roaming the steppes would still be related in stories, with the presumption they had left the world of the mortal for the place the ancestors of all life dwelt in. As time passed, hunters did not stalk prey on foot, but sped on horseback after their quarry. If you have ever had the raw delight of allowing a horse to gallop across a flat landscape, you will know that you feel the most complete sense of freedom, as though you are riding the wind itself. This sense of elation would likely be transferred into otherworldly experiences under the influence of hallucinogenic substances, which we know was an important part of the shamanic practice of steppes tribes such as the Pazyryk peoples of the Iron Age, c 500 BC.
In this altered, shamanic state, the hunted Siberian Red Deer would likely be considered a smaller, earthly manifestation of the remembered ancestor of deer, the Megaloceros. Everything is better, more ‘real’ and more heroic on the Other Side, as postulated by Plato in his Theory of Forms. Horses chosen for sacrifice, which had once run down the lesser, extant species of deer, were making a transition to the heavenly pastures, sharing qualities with the hunted while still being part of the hunting team. Perhaps this is why the unfortunate horses were decked out in the crown of tines to represent the formidable giant deer which once shared those wild plateaux and plains. It is said in many religions of the past that things remembered never actually die.
Just like the sinuous tattoos of the ice mummies are interconnected, and always looking back at each other, so too archaeology, palaeoenvironmentology and palaeontology are interlinked. Papers such as van der Plicht et al’s new dates for the last of the Siberian Megaloceros allow us to look at later cultural practices with questions and potentials. For those of us who love teasing out the knotted wool of archaeology, such new findings of refugia which were shared with humans can only offer more questions and more exploration of that time when giants did indeed walk the earth.
Written by Rena Maguire (@JustRena)
The research and radiocarbon dating that inspired this post is from van der Plicht, J, et al. (2015), ‘New Holocene refugia of giant deer (Megaloceros giganteus Blum.) in Siberia: updated extinction patterns’ Quaternary Science Reviews, 2015, 114. [Full article]
A nice overview of the Giant Deer (Megaloceros giganteus) can be read in An Elk that wasn’t an elk.
Further Reading:
Bayarsaikhan, J. (2005), ‘Shamanistic elements in Mongolian deer stone art’. In: Fitzhugh, W, Bayarsaikhan, J. & Marsh, P.(eds), The Deer Stone Project: Anthropological Studies in Mongolia 2002–2004. Washington, D.C.: Smithsonian Institution, 41–53. [Full article]
Brentjes, B., (2000), ‘Animal Style’ and shamanism: Problems of pictoral tradition in Northern and Central Asia’. In: J. Davis-Kimball, E. Murphy, L. Koryakova and L.T. Yablonsky, eds. Kurgans, Ritual Sites, and Settlements Eurasian Bronze and Iron Age, BAR International Series 870. Oxford: Archaeopress. 259-268. [Full article]
Brück, J., (1999), ‘Ritual and rationality: Some problems of interpretation in European archaeology’. European Journal of Archaeology. 2.(3). pp.313-344. [Abstract only]
Gryaznov, M.P., (1950), Pervyi Pazyrykskii Kurgan. (First Pazyryk Kurgan.) ) St. Petersburg:Hermitage. (Russian.)
Gryaznov, M.P., (1969), The Ancient Civilization of Southern Siberia. J. Hogarth, trans. Geneva: Nagel. [Book]
Kuzin, Y & Orlova, L. (1998), ‘Radiocarbon chronology of the Siberian Palaeolithic’. Journal of World Prehistory. 12. (1). pp.1-53. [Full article]
Mallory, J. P. (1981), ‘Ritual treatment of the horse in early Kurgan cultures’. Journal of Indo European Studies. 9. (3-4). pp.205-227.
Outram, A. et al. (2009). ‘The earliest horse harnessing and milking.’ Science 323.(5919). pp.1332-1335. [Full article]
Ripinsky-Naxon, M., (1993). The Nature of Shamanism: Substance and Function of a Religious Metaphor. Albany: State of New York University Press. [Book]
Rudenko S. I., 1970 [1953]. Frozen Tombs of Siberia. The Pazyryk Burials of Iron-Age Horsemen. M.W. Thompson, trans. Berkeley and Los Angeles: University of California Press. [Book]
Samashev, Z., (2007), ‘Culture of the nomadic elite of Kazakhstan’s Altai Region (based on materials for the Berel necropolis.)’ In: C. Chang and K.S. Guroff, eds. Of Gold and Grass: Nomads of Kazakhstan, pp.35-44. [Book]
Samashev, Z., et al. (1999). Berel mounds [online]. Kumbez. Available at: http://www.lorton.com/~kumbez/archelogy_2_eng.htm
Shanks, M. & Tilley, C., (1982). ‘Ideology, symbolic power and ritual communication: A reinterpretation of Neolithic mortuary practices’. In: I. Hodder, ed. Symbolic and Structural Archaeology. Cambridge: Cambridge University Press. pp.129-154. [Book]
Sinor, D., (1998), ‘The myth of languages and the language of myth’. In: V. H. Mair, ed. The Bronze Age and Early Iron Age Peoples of Eastern Central Asia. Philadelphia: Institute for the Study of Man and University of Pennsylvania Museum. pp.729-745. [Book]
Stuart, A.J., Kosintsev, P.A., Higham, T.F.G., Lister, A.M., 2004. ‘Pleistocene to Holocene extinction dynamics in giant deer and woolly mammoth’. Nature 431. 684-689. [Abstract only]
Vislobokova, I. A., (2012). ‘Giant deer: origin, evolution, role in the biosphere’. Palaeontology Journal. 46. pp.643-775. [Full article]
Vitebsky, P., (2005). The Reindeer People: Living with Animals and Spirits in Siberia. Boston:Houghton Mifflin. [Book]
Volkov, V. (1981), Olennyie Kamni Mongolii (Deer Stones of Mongolia). Ulaanbaatar: Academiya Nauk.
http://siberiantimes.com/science/casestudy/features/iconic-2500-year-old-siberian-princess-died-from-breast-cancer-reveals-unique-mri-scan/